 Click to Print Adobe PDF
Click to Print Adobe PDF
Spring 2009 - Vol.4, No.1
Surgery for Diseases of the Vitreous and Retina
Roy D. Brod, M.D.
Associate Clinical Professor of Ophthalmology
Hershey / Penn State University School of Medicine
|
 |
Abstract
The discovery in 1962 that the vitreous gel can be safely removed from the eye has led to the development of a surgical procedure that has restored or improved vision and prevented blindness in countless patients around the world. The field of vitreoretinal surgery has expanded enormously over the past 47 years and it is now an established subspecialty of ophthalmology, usually requiring a two-year fellowship after completion of ophthalmology residency training. Our understanding of the pathobiology and pathophysiology of vitreoretinal diseases combined with technological advancements in instrumentation for vitreoretinal surgery has led to many new indications for vitrectomy surgery since the technique was first performed in the 1960s. Not only can opaque vitreous, usually due to hemorrhage, be removed to restore vision, but also scar tissue can be peeled from the surface of the retina, retinal detachments can be repaired, macular holes can be closed, and even large vitreous opacities causing bothersome shadows on the retina and the perception of floaters can be successfully removed using newer and safer vitrectomy techniques. This review will discuss the history, techniques, indications, and complications of vitreoretinal surgery, as well as pertinent ocular anatomy.
Introduction
The vitreous can cause deterioration of visual acuity as a result of many disease processes such as diabetic hemorrhage, but age-related changes and floaters are inevitable and common. Before discussing the diverse disorders of the vitreous that can lead to surgery, some background in ocular anatomy[1] and physiology is necessary. We will then discuss the evolution of modern techniques for vitreoretinal surgery, its indications, and its outcomes.
Anatomy of the Eye
The vitreous is a relatively clear, gel-like structure that fills the space between the lens and the retina [Fig. 1]. It has a volume of approximately 4 ml in the adult eye or approximately 80% of the volume of the entire eye, and is composed mainly of collagen and hyaluronic acid that interact in interpenetrating networks. The collagen fibrils give the vitreous its solid structure and the hyaluronic acid contributes to its volume.[2] The vitreous has two main portions, a central area called the vitreous body and a thin outer layer called the vitreous cortex that is attached to the retina and posterior surface of the lens.
An important anatomical reference point in the eye is the limbus, where the cornea meets the sclera. In the adult eye, the anterior extent of the retina (ora serrata) is located approximately 6-8 mm posterior to the limbus (closer on the nasal side of the eye). Thus, any incision made more than 6-8 mm posterior to the limbus will likely perforate the retina and potentially lead to a retinal detachment. Furthermore, approximately 2 mm posterior to the limbus is the pars plicata, a highly vascular zone which contains the ciliary processes that produce the aqueous humor. An incision through the pars plicata is associated with a markedly increased risk of bleeding. Fortunately, just posterior to the pars plicata is the pars plana, which extends 4-6 mm from the posterior edge of the pars plicata to the ora serrata where the retina begins. The pars plana is the optimal location for entry into the eye, because it is not very vascular, and is still anterior to where the retina begins. The incisions for vitrectomy are usually made through the pars plana 3 to 4 mm posterior to the limbus, which explains the correct terminology for this procedure: a pars plana vitrectomy.
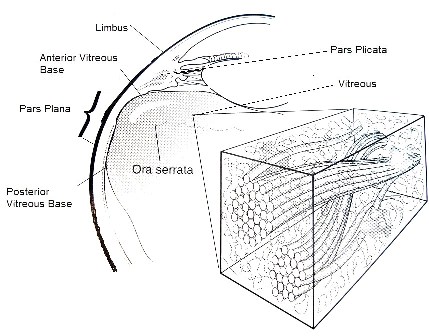 |
Figure 1. The ocular anatomy important for understanding pars plana vitreous surgery.
Source: American Medical Association, Copyright 2002. |
Evolution of Vitrectomy Instruments and Techniques
Vitreoretinal surgery has undergone considerable evolution since 1962, when Kasner introduced the concept of removing the vitreous gel from the eye as a therapeutic modality.[3] Operating on a traumatized eye, he intentionally removed the vitreous through a large corneal wound. This "open sky" technique was later utilized to remove opacities from the vitreous in eyes that would have otherwise remained blind, usually from vitreous hemorrhage. Kasner's open approach paved the way to a safer and more effective closed system technique for removing the vitreous.
Dr. Robert Machemer pioneered this approach and performed the first closed system vitrectomy in 1972, using a prototype instrument called the VISC - vitreous infusion suction cutter.[4] (Simply aspirating vitreous was not feasible due to its high viscosity as well as the risk of creating retinal tears. Retinal detachment can result if retinal traction is induced by aspirating vitreous without simultaneously cutting.) By placing the VISC instrument through an incision in the pars plana region of the eye, Machemer was able to remove the vitreous gel with a cutting and sucking action, while at the same time maintaining pressure in the eye by simultaneously infusing a physiologic saline solution manually with a syringe. The prototype cutting mechanism utilized a rotating blade that cut small fragments of vitreous that were then aspirated into the port of the instrument. In order to improve visualization into the eye, a fiber-optic light sleeve was added to the instrument. An operating microscope was later utilized to obtain adequate magnification during the procedure.
This early prototypical instrument with the attached light sleeve was relatively thick, necessitating a 3.5 mm incision in the pars plana. Although this incision was much smaller than the original corneal "open sky" incision, it was still a relatively large incision to make in the pars plana. Thus, although this early technology was very effective in removing the vitreous, a number of modifications have been made over the ensuing years.
To reduce the size of the instrument and the incision needed to insert it, a divided instrument system was developed.[5] Instead of using a single instrument for aspiration, cutting, infusion, and illumination, it was divided into three separate instruments, thus reducing the size of each instrument to a diameter of approximately 1 mm. The weight of each instrument was also significantly reduced, enabling better control of the eye by the surgeon. In addition, the smaller pars plana incision reduced the risk of wound-related complications, including retinal tears and detachments due to vitreous incarceration in the wound. The cannula to infuse physiologic saline solution as the vitreous is removed was placed at a separate site and is usually sutured in place. The two other 1mm scleral incisions made into the eye are for the fiberoptic light probe and the vitreous cutter/aspirator (Figs. 2,3). Instead of one instrument in one hand, the surgeon now has two hands (the vitreous cutter in one hand and the illumination probe in the other), which provides much better control of the eye and also markedly improved visualization. To enhance the focus on structures in the posterior portions of the eye, including the retina, a plano-concave contact lens is placed on the corneal surface, which neutralizes the refractive power of the eye.
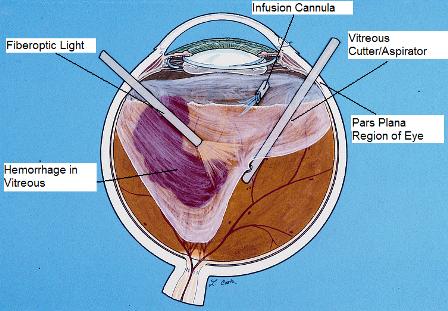 |
| Figure 2. Diagram depicting a 3-port closed system pars plana vitrectomy in an eye with a vitreous hemorrhage. Note the infusion cannula, fiber-optic light pipe, and vitreous cutter inserted into the eye through the pars plana. |
 |
| Figure 3. Photograph of an eye prepped and undergoing a 3-port vitrectomy procedure. |
This brings us to the current 3-port technique utilized in modern vitreoretinal surgery. The vitreous cutter is now driven pneumatically with pressurized gas which drives a reciprocating or linear cutter that cuts the vitreous like a guillotine at speeds up to 2500 cuts per minute as it is aspirated into the port of the cutter. This high-speed cutter significantly reduces vitreoretinal traction that - as noted earlier - can lead to iatrogenic retinal tears. An infusion pump, also driven by gas pressure, infuses a balanced salt solution into the eye at a pre-set constant pressure. During the operation, the pressure in the eye can be elevated instantly if necessary to help control intra-operative bleeding. The infusion pump, as well as the light source and pneumatic driver for the vitreous cutter/aspirator, is contained in a computerized console placed adjacent to the operating table (Fig. 4).
 |
| Figure 4. Photograph of the computerized vitrectomy console that contains the driver for the vitreous cutter, the fiber optic light source and the infusion pump. |
As experience with the surgical technique improved, more indications for vitrectomy evolved. This necessitated development of accessory instrumentation, such as intraocular microscissors, forceps, soft tip aspiration cannulas, vitreoretinal picks, and even an intraocular laser probe. To better visualize the peripheral retina, additional contact lenses with different magnifications and powers were introduced. Eventually a wide-field viewing system that did not require placement of a contact lens on the eye was developed and is now commonly used for better visualization of the peripheral vitreous and retina.
Although the 3-port divided vitrectomy system enabled instruments with much smaller diameters (approximately 1 mm), each pars plana wound still required a suture for a watertight closure. This was usually a biodegradable suture, but it stimulated ocular surface inflammation and was a source of postoperative discomfort. In addition, even though the 1 mm wound is much smaller than the early 3.5 mm wound, it still allowed some vitreous to prolapse into the wound as instruments were removed from the eye, leading to postoperative retinal tears in approximately 5-10% of cases.[6]
In order to further refine the technique and reduce postoperative inflammation and operative time, smaller diameter instruments were developed and first utilized clinically in 2005. These instruments were slimmed down to 25 gauge or approximately 0.5 mm in diameter and were inserted through a pre-placed cannula that was inserted with a sharp trocar. The cannula system prevented enlargement of the wound from repeated entry and withdrawal of instruments, and reduced the incidence of vitreous prolapse during the procedure. In addition, the 25 gauge incisions were self-sealing and sutures were not required. Not only did this reduce postoperative inflammation and discomfort, as well as the incidence of retinal tears, but it also reduced operating time and cost. The trade-off, however, was greater flexibility of the thinner instruments, which diminished the surgeon's ability to rotate the eye into different positions without bending them. Also, the smaller diameter reduced flow and increased the time needed to remove the vitreous.[7]As a result, 23-gauge instruments were developed which are approximately 0.65 mm in diameter and also utilize a cannula system with self-sealing incisions. Not only are these stiffer than the 25-gauge instruments, but they carry higher flow and require less time to remove the vitreous. Both 25 and 23 gauge systems are utilized today; the choice depends mainly on the surgeon's preference and the particular surgical indication.[8]
Changes in the Vitreous and Their Consequences
With age, the vitreous gel becomes more liquefied, which causes optically empty spaces to develop, surrounded by thick fibrillar condensations.[9] These fibrillar condensations scatter incident light and may produce shadows on the retina, perceived by an individual as floaters. With time and increasing age, the liquefied cavities coalesce. Eventually a break or hole occurs in the posterior vitreous cortex and liquid vitreous pours through the break, getting underneath the cortical vitreous and separating the surrounding cortical vitreous from the retina. This is usually a sudden event and it rapidly progresses from posterior to anterior where it stops at a region called the vitreous base, approximately 1.8-3.0 mm posterior to the ora serrata (Fig. 1). At this site the vitreous is markedly adherent to the peripheral retina and pars plana.
This age-related, naturally occurring event is called a posterior vitreous detachment or PVD. When it occurs, the patient usually notes an increase in floaters or the sudden onset of new floaters. In some cases, vertically oriented flashes of light are noticed in the temporal visual field of the eye. If the vitreous is abnormally adherent to the retina as it is separating from the retina, a retinal tear may develop, and there may be an associated vitreous hemorrhage. In some cases, a vitreous hemorrhage will develop without a tear, as the vitreous pulls on a retinal vessel without actually pulling on and tearing the retina.
All patients should be warned to immediately report symptoms of new or increased floaters especially if associated with flashing lights, and to seek ophthalmologic attention as soon as possible to rule out a retinal tear.[10] Retinal tears occur in approximately 15% of patients who experience symptoms of a PVD. That percentage rises to about 30% if there is an associated vitreous hemorrhage, and it approaches 100% if there are pigment granules in the vitreous.[11] Although in most cases a retinal tear cannot be prevented, if it is treated expeditiously it is possible to prevent a resulting retinal detachment. In the majority of patients, the increase in number and size of vitreous floaters is self-limited and either improves spontaneously or the patient becomes less aware as the brain begins to ignore them. In some cases, very large and numerous opacities can be persistent and become visually significant. The symptoms are due not only to the perception of the vitreous opacities themselves, but also of the increased glare that results from the scattering of incident light.
Indications for Vitreoretinal Surgery
The indications for vitrectomy have evolved in conjunction with advances in surgical skill and technology and our understanding of vitreoretinal pathology (Table 1). The initial sole indication for vitrectomy was to remove densely opacified vitreous that obscured vision. This problem usually resulted from hemorrhage into the vitreous, often secondary to complications of diabetic retinopathy. Prior to the development of vitreoretinal surgery, such patients often remained blind, but now the vision in these eyes can usually be restored.[12] In the category of "other vitreous opacities" that can now be removed is inflammatory material that results from either an intraocular infection or a sterile inflammatory process. A neoplastic infiltration of the vitreous, resulting from a lymphomatous infiltrate, can mimic an inflammatory process. In such cases, the vitrectomy is used for diagnostic as well as therapeutic purposes.
Table 1: Indications for Vitrectomy
- Vitreous Hemorrhage (diabetic and other causes)
- Vitreous Inflammation
- Non-infectious
- Infectious (Endophthalmitis)
- Vitreous floaters (Severe and Visually significant)
- Retinal detachment - repair
- Macular holes - repair
- Retinal scar tissue - removal (idiopathic, diabetic related, associated with retinal detachment)
- Retained cataract fragments after cataract surgery - removal
- Dislocated lens implants - retrieval
- Retinopathy of prematurity - repair complications
- Refractory diabetic macular edema – membrane peeling and vitrectomy
- Severe ocular trauma - repair and removal of intraocular foreign bodies
- Indeterminate diagnosis - obtain vitreous specimen
|
Initially, only severe vitreous opacities that resulted in marked visual loss were considered for vitrectomy surgery, but as the technology and surgical skill improved and the risk of complications fell, less severe cases were accepted. An example of an evolving indication for vitrectomy surgery is large vitreous floaters, usually in patients with a previous posterior vitreous detachment. Although these vitreous opacities do not cause severe visual loss or blindness and can be associated with excellent visual acuity, they can result in extremely bothersome symptoms that in some cases are debilitating to the patient. They are particularly annoying to those who work with a computer, where they can be highly visible and distracting against the white screen. Although the YAG laser has been advocated as a therapy for such vitreous opacities the results are variable at best and the YAG laser technique for treating vitreous opacities is not often successful.[13] With the advent of small gauge, sutureless vitrectomy, these large vitreous opacities can be removed successfully in selected symptomatic patients with relatively little intraoperative risk and minimal postoperative discomfort. [14][15]
Besides removal of vitreous opacities, surgery can remove scar tissue adherent to the surface of the retina whether due to diabetes, an associated retinal detachment, or idiopathic causes. Small vitreoretinal forceps have been developed to grasp and peel the scar tissue membranes from the surface of the retina. In certain cases where the membranes cannot be peeled, a small scissors can be inserted into the eye to cut or segment the membrane, relieving retinal traction in this way. Retinal detachments can also be repaired using vitrectomy techniques. This procedure is combined with an infusion of a long acting intraocular gas to tamponade the retina, followed by intraoperative laser photocoagulation.[16] Other indications for vitreoretinal surgery include repair of macular holes; removal of dislocated cataract fragments or intraocular lens implants; repair of severely traumatized eyes and removal of intraocular foreign bodies; removal of sub-retinal hemorrhage in cases of macular degeneration; and treatment of diabetic macular edema in eyes unresponsive to less invasive treatments.[17]
Complications
Complications of vitreoretinal surgery have become much less frequent as the technique, technology, and surgical skills have evolved, but certain complications may still occur (Table 2).[18] One of the more common complications, or - more precisely - side effects, of vitrectomy is the progression of a cataract in a patient who still has their natural lens (phakic).[19] This complication is obviously not possible if the lens has already been removed (pseudophakic). This progression of an already existing cataract occurs relatively commonly after vitrectomy for any indication, but the incidence is higher when a gas bubble is placed in the eye during surgery. In addition, the progression of a cataract increases with the increasing age of the patient at the time the vitrectomy is performed. The rate of cataract progression is not increased in patients younger than 50 years at the time of vitrectomy. It is now evident that the increased rate of cataract formation after vitrectomy is due to increased oxygen tension in the vitreous cavity of an eye that has undergone vitrectomy surgery, which explains the association with the aforementioned use of a gas bubble.
Table 2: Complications of Vitrectomy
- Cataract progression
- Infection (endophthalmitis)
- Retinal tear
- Retinal detachment
- Hypotony
- Glaucoma
- Vitreous cavity hemorrhage
- Suprachoroidal hemorrhage
|
Other potential complications include postoperative intraocular infection (endophthalmitis),[20] a rare but serious complication that occurs in less than one in 1,000 cases. A postoperative retinal tear or detachment can also occur. Actual retinal detachments have been reported in 5%-15% of cases, but the incidence appears to be lower with the newer small gauge vitrectomy surgery.[21] Statistics comparing the incidence of this complication among 20 gauge and 23 or 25 gauge procedures will be forthcoming as data are collected. Postoperative vitreous hemorrhages, particularly following vitrectomy for complications of diabetes, may occur. One complication that may be more frequent with the smaller gauge surgical technique is postoperative hypotony, or low intraocular pressure.[22] This problem is usually seen on the first postoperative day and is generally self-limited, though occasionally it persists due to a leak in one of the incisions that fails to self-seal, and requires a return to the operating room for placement of a suture.
In general, serious complications are infrequent in modern vitreoretinal surgery and in the vast majority of cases a successful outcome is obtained.
References
[1] Wolfe E. In Warick R (ed). Anatomy of the Eye and the Orbit, 7th ed. Philadelphia, WB Saunders Co, 1976.
[2] Sebag J, Balazs A. Morphology and ultrastructure of human vitreous fibers. Invest Ophthalmol and Vis Sci 1989;30:187-191.
[3] Kasner D. A new approach to the management of the vitreous. Highlights Ophthalmol 1968;11:304.
[4] Machemer R, Buettner H, Norton EWD. Vitrectomy: a pars plana approach. Trans Am Acad Ophthalmol Otolaryngol 1971;75:813.
[5] O'Malley C, Heintz RM. Vitrectomy with an alternative instrument system. Ann Ophthalmol 1975; 7:585
[6] Scartozzi R, Bessa AS, Gupta OP, Regillo CD. Intraoperative sclerotomy-related retinal breaks for macular surgery, 20-vs-25 gauge vitrectomy systems. Am J Ophthalmol 2007;143;155-156
[7] Chen E. 25-gauge transconjunctival sutureless vitrectomy. Curr Opin Ophthalmol 2007;18:188-193
[8] Williams GA. 25, 23, or 20 Gauge instrumentation for vitreous surgery. Eye 2008; 22:1263-1266
[9] Sebag. Age-related changes in human vitreous structure. Albrecht Von Graefes Arch Clin Exp Ophthalmol 1987;225:89-93
[10] Margo CE, Harmon LE. Posterior vitreous detachment. How to approach sudden- onset floaters and flashing lights. Postgrad Med 2005;117:37-42
[11] Brod RD, Lightman DA, Packer AJ, Saras HP. Correlation between vitreous pigment granules and retinal breaks in eyes with acute posterior vitreous detachment. Ophthalmology 1991;:98:1366-1369.
[12] Mason JO lll, Colagross CT,Vail R. Diabetic vitrectomy: risks, prognosis, future trends. Curr Opin Ophthalmol 2006;17:281-285.
[13] Delaney YM, Oyinloye A, Benjamin L. Nd:YAG vitreolysis and pars plana vitrectomy: surgical treatment for vitreous floaters. Eye 2002; 16:21-26.
[14] Schiff WM, Chang S, Mandava N, Barile GR. Pars plana vitrectomy for persistent, visually significant vitreous opacities. Retina 2000;20:591-596.
[15] Weber-Varszegi V, Senn P, Becht CN, Schmid MK. "Floaterectomy"- pars plana vitrectomy for vitreous opacities. Klin Monatsbl Augenheilkd 2008; 225:366-369.
[16] Miller DM, Riemann CD, Foster RE, Peterson MR. Primary repair of retinal detachment with 25-guage pars plana vitrectomy. Retina 2008; 28:931-936.
[17] Hartley KL, Smiddy WE, Flynn HW JR, Murray TG. Pars plana vitrectomy with membrane peeling for diabetic macular edema. Retina 2008;28:410-414.
[18] Gupta OP, Weichel ED, Regillo CD, Fineman MS, Kaiser RS, Ho AC, McNamara JA, Vander JE. Postoperative complications associated with 25-guage pars plana vitrectomy Ophthalmic Surg Lasers Imaging 2007 ;38:270-5.
[19] Thompson JT. The role of patient age and intraocular gas use in cataract progression after vitrectomy for macular holes and epiretinal membranes. Am J Ophthalmol 2004; 137:250-257
[20] Elfig CW, Scott IU, Flynn HW Jr, Smiddy WE, Newton J. Endophthalmitis after pars plana vitrectomy: incidence, causative organisms, and visual acuity outcomes. Am J Ophthalmol 2004;138:799-802.
[21] Moore JK, Kitchens JW, Smiddy WE, Mavrofrides EC, Gregorio G. Retinal breaks observed during pars plana vitrectomy Am J Ophthalmol 2007 25:32-36.
[22] Hsu J, Chen E, Gupta O, Fineman MS, Garg SJ, Regillo CD. Hypotony after 25-gauge vitrectomy using oblique versus direct cannula insertions in fluid-filled eyes. Retina 2008;28:937-940.
Roy D. Brod, M.D.
2150 Harrisburg Pike – Suite 270
Lancaster, PA 17604
717-399-8790
roydbrodmd@dejazzd.com
Neither Dr. Brod nor any member of his immediate family have any relevant relationships with any corporate organizations associated with the manufacture, license, sale, distribution or promotion of a drug or device to disclose.