Summer 2022 - Vol. 17, No. 1
SCIENTIFIC REPORT
Lipid Update
What Should Be Done About Residual Triglycerides?
Joette C. Hughes, MSN, CRNP, CLS
Cardiovascular Nurse Practitioner
The Heart Group of Lancaster General Health
Non-high-density lipoproteins (non-HDL) is a measurement of the sum of low-density lipoproteins (LDL), intermediate-density lipoproteins (IDL), very low-density lipoproteins (VLDL), chylomicron remnants, and lipoprotein (a). Triglycerides are a portion of non-HDL, represented in VLDL and chylomicron remnants.
Triglyceride-rich lipoproteins (TRLs) are involved in the initiation and propagation of atherosclerosis through their invasion in the arterial wall.
1 They further activate inflammation in the arterial wall through the secretion of tumor necrosis factor-β and interleukin 1-β, thought to set off the process of atherogenesis via endothelial dysfunction.
2
Recent guidelines emphasize that while the focus of atherosclerotic cardiovascular disease (ASCVD) prevention should remain on LDL, we should not ignore elevated levels of apolipoprotein B (ApoB)-containing cholesterol — of which non-HDL is a surrogate marker — and triglycerides. Retrospective data from the Framingham Offspring study and Atherosclerosis Risk in Communities study showed a linear relationship between average triglyceride level and ASCVD risk, even in those with triglyceride levels less than 150 mg/dL.
3 Thus, it would seem, lower triglyceride levels are associated with decreased coronary risk, and there does not appear to be a “safe” level.
Further, if a patient is considered “at risk” for ASCVD events and treated with statins to push the LDL to levels <70 mg/dL, individuals with triglycerides ≥150 mg/dL still have a 41% higher risk of coronary events.
4 Thus, non-HDL should be a co-target for both primary and secondary prevention.
When lipid levels are checked in the fasting state, they can provide an accurate measurement of triglycerides, especially if there is a family history of premature ASCVD or a genetic cholesterol disorder. Optimal fasting triglyceride levels should be <100 mg/dL.
4 In general, levels between 150 and 199 mg/dL are borderline high, 200 to 499 mg/dL high, and ≥500 very high. The risk for pancreatitis increases with levels above 500 mg/dL, with substantial risk ≥1000 mg/dL.
5
PRIMARY CAUSES
Being overweight (BMI 25-29 kg/m
2) or obese (BMI ≥30 kg/m
2) raises the chance of having high triglycerides and elevated non-HDL. Dietary intake of sugar or simple, processed carbohydrates, as well as fats, can raise the levels of triglycerides. Thus, it is essential to tease out what may be considered innocuous dietary habits (e.g., use of coffee creamer), but may impart significant risk toward dyslipidemia. Oils or spreads used for cooking, specifically coconut oil or butter, can place a high burden through saturated fats.
There has been much confusion in the media about healthy behaviors in dietary preparation; providers must work to clear up misconceptions. Regular use of alcohol may exacerbate hypertriglyceridemia, but a ketogenic diet — one high in fat, but low in carbohydrates — can successfully reduce weight and improve cholesterol levels. However, if done inappropriately, this diet pattern may contribute to dyslipidemia. Further, a ketogenic diet should be avoided in those who have genetically derived cholesterol problems, such as those with familial hypercholesterolemia (FH), an inherited disorder of LDL common in the population of Lancaster County.
SECONDARY CAUSES AND GENETICS
It is widely recognized that many disease processes and medicines can contribute to increased levels of triglycerides (see Table 1). Genetics are also an important consideration.
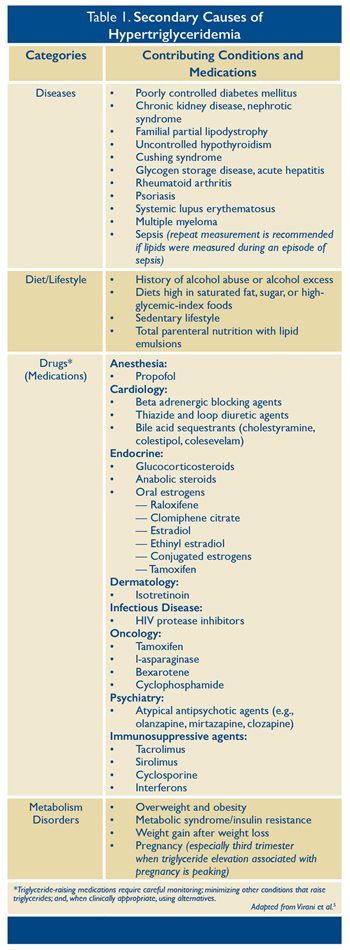
The most common monogenic mutation causing hypertriglyceridemia is Familial Chylomicronemia Syndrome (FCS), an autosomal recessive disorder characterized by triglyceride levels >99th percentile. FCS results from mutations causing the ineffectiveness of lipoprotein lipase or alteration of ApoC-II, leading to the inability to break down triglycerides.
6 Patients with FCS have very high levels of chylomicrons, and when diagnosed may have triglyceride levels above 880 mg/dL despite medication treatment.
A hallmark of FCS is recurrent episodes of pancreatitis, although these patients do not have a higher incidence of ASCVD. This disorder is rare, with an incidence of approximately one in a million, yet accounts for 95% of monogenic mutations. The remainder of patients with mutations have errors in genes for ApoC-II, GPIHBP1, ApoA-V, and LMF1.
Other genetic causes of hypertriglyceridemia that are recurrent fall under the umbrella of Multifactorial Chylomicronemia Syndrome, or MCS, a polygenic hypertriglyceridemia. Familial Combined Hyperlipidemia (FCH) is characterized by high triglycerides with or without high LDL. These patients have a significant amount of ApoB cholesterol particles, generally ≥130 mg/dL, which can be distributed between triglyceride and LDL burden. This can be difficult to diagnose as LDL levels can vary, although triglycerides tend to remain high. In most of these cases, the true LDL cannot be evaluated until triglyceride levels improve.
Interestingly, while FH is very common among the Pennsylvania Dutch population, there is also a mutation in the gene for ApoC-III in the Amish community that leads to abnormally low levels of triglycerides. ApoC-III acts primarily as an inhibitor of lipoprotein lipase, thereby raising levels of triglycerides.
Finally, Familial Dysbetalipoproteinemia is characterized by mutations in ApoE. This exists in about one in 10,000 people, with elevated IDL and chylomicron remnants. Those afflicted will have high levels of cholesterol and triglycerides with low LDL. Patients may demonstrate eruptive xanthomas and are at increased risk for ASCVD.
TREATMENT
Lifestyle modification is the first step in the treatment of any cholesterol disorder. Clinicians should suggest that patients bring a three-day food diary to expedite review.
5 Dietary recommendations depend on triglyceride levels, with stricter recommendations reserved for patients with the highest levels and thus the most risk (see Table 2). These include attention to fat, alcohol, and carbohydrate intake.

Along with this recommendation, exercise in the form of at least 150 minutes of moderate or 75 minutes of vigorous exercise per week should be encouraged. Additionally, patients with BMI >25% may find that weight loss of 5% to 10% of total body weight can contribute to a 20% or more reduction in triglyceride levels.
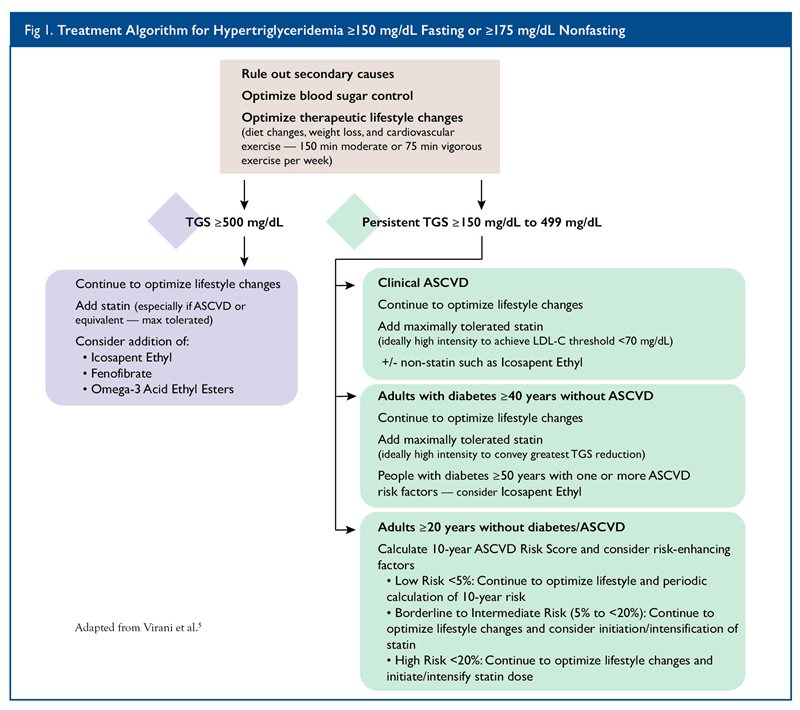
In 2021, the American College of Cardiology (ACC) revealed its Expert Decision Pathway on the Management of ASCVD Risk Reduction in Patients with Persistent Hypertriglyceridemia.
5 Statins remain the primary treatment for those with ASCVD and can lower triglyceride levels reasonably. For those with triglyceride levels above 500 mg/ dL, triglycerides become a primary target to prevent complications such as pancreatitis. Clinicians may still utilize statins first, especially in the presence of ASCVD; otherwise, these guidelines suggest that if a patient’s cardiovascular risk is low, the use of icosapent ethyl (
Vascepa1), fenofibrate, or omega-3 acid ethyl esters (
Lovaza1) should be considered.
Consistent with the 2018 guidelines, the 2021 Decision Pathway indicates that if triglyceride levels are below 500 mg/dL, treatment should focus on LDL first, then on non-HDL. Further, for those patients at increased ASCVD risk who have attained an LDL goal, yet have residual triglycerides between 150 mg/ dL and 499 mg/dL, clinicians should consider the addition of icosapent ethyl.
Statins
In addition to lowering the risk for ASCVD and major adverse cardiac events (MACE), statins can induce a 10% to 30% reduction in triglycerides dependent upon medication selected and dose. In adults with ASCVD and hypertriglyceridemia, or those 40 and above with diabetes and hypertriglyceridemia, maximal statin therapy is the primary treatment recommendation in addition to diet and lifestyle changes.
5 For those 20 years old or greater with persistent hypertriglyceridemia who will not become pregnant, statin therapy may be considered after lifestyle changes, elimination of secondary causes, and calculation of the 10-year ASCVD risk assessment tool.
Fibrates
Fibrates include gemfibrozil and fenofibrate. The VA-HIT trial studied gemfibrozil in more than 2,500 men with ASCVD, though most were not on statin therapy. While a benefit was seen, the sample size was small and there was no extensive use of statin therapy. No studies have shown a benefit of gemfibrozil in addition to statin therapy. More notable is the potential for significant interaction with statin therapy, and this combination should be avoided.
Fenofibrate has been studied in several trials, including the ACCORD trial with more than 5,000 people with diabetes on baseline simvastatin. There was no significant benefit in cardiovascular outcomes noted. The FIELD trial studied nearly 10,000 people with diabetes treated with fenofibrate who were not on baseline statin therapy. Though there was no statistical difference in coronary events compared to placebo, non-fatal myocardial infarction decreased by 24%. Most notable was improvement in albuminuria and retinopathy, suggesting benefit from microvascular complications.
7
Omega-3 Fatty Acids
While none of the recent major studies (VITAL, ASCEND, OMEMI, and STRENGTH) using combi. Nation omega-3 fatty acids showed any clinical benefit or reduction in MACE, combination omega-3 acids are an option to treat triglycerides above 500 mg/dL. Regarding a derivative, icosapent ethyl, the results are more encouraging.
The 2019 Reduce-It trial examined the use of 2 grams twice daily (purified EPA only —
Vascepa1) versus placebo in over 8,000 patients with existing cardiovascular disease or diabetes and two additional risk factors. The baseline triglyceride levels were between 135 mg/ dL and 499 mg/dL, and patients were all given statin and demonstrated median LDL levels of 75 mg/dL.
Over 4.9 years of follow-up, there was a reduction in MACE by 25% (number needed to treat [NNT] = 28). Cardiovascular death improved by 20%, ischemic stroke by 29%, and improvement was seen in rates of hospitalization from heart failure as well.
5
Side effects did include a higher risk for atrial fibrillation (5.3% vs. 3.9%), peripheral edema (6.5% vs. 5.0%), and adverse serious bleeding (2.7% vs. 2.1%), although there was no fatal bleeding. In general, anemia and gastrointestinal side effects were better in the treatment group. Thus, in 2019 the National Lipid Association endorsed the use of icosapent ethyl in statin-resistant hypertriglyceridemia, especially in patients with high or very high risk for clinical ASCVD.
8
Over-the-counter fish oil products are not recommended due to the differences in concentration of omega-3 fatty acids and purity, as well as the number of pills needed and gastrointestinal side effects.
Niacin
Niacin is vitamin B-3, though for cholesterol treatment it is administered in levels much higher than dietary consumption — 500 mg or higher.
While early studies with niacin alone showed cardiovascular benefit in secondary prevention, the results of the AIM-HIGH study (2011) for those with existing ASCVD on baseline statin therapy showed no benefit. After three years, the study was stopped early due to futility. Subsequently, in the HPS2-THRIVE trial (2014), extended-release niacin was studied in combination with laropiprant, an agent added to prevent flushing, in those with baseline ASCVD on statin. This trial, too, was stopped early, due to a high level of major adverse events. Niacin can cause flushing, hepatotoxicity, insulin resistance, and gout flares.
9
In 2016, the FDA stated the risks outweighed the benefits in combining extended-release niacin with statin medication. Niacin may still be considered as monotherapy for severe hypertriglyceridemia (≥1000 mg/dL), but caution should be used and side effects considered. The 2018 ACC/AHA guidelines do not endorse the use of niacin in combination with statin therapy.
10
 CLINICAL TRIALS IN PROGRESS
CLINICAL TRIALS IN PROGRESS
There has been an advent of many new classifications of medicine treatment, including monoclonal antibodies, small interfering RNA, and antisense technology. Monoclonal antibodies are laboratory-made antibodies, similar to those occurring naturally, which can target a single site for medication benefit. Small interfering RNA technology utilizes a small double-stranded RNA molecule that interferes with transcription or production of a gene or protein. Antisense oligonucleotide drugs are single-stranded RNA molecules that interfere with the transcription of a protein. Several phase 2 and 3 studies are currently underway.
Three companies have three different technologies against ANGPTL3, in which interference can improve both triglycerides and total cholesterol levels. These drugs include:
- evinacumab (Evkeeza1), a monoclonal antibody currently approved for LDL treatment in homozygous FH (mutations in both alleles of genes responsible for LDL processing);
- ANGPTL3-LRx, an antisense drug; and
- ARO-ANG3, an siRNA drug.
Two drugs targeting ApoC-III, which can improve triglyceride levels by increasing lipoprotein lipase levels, are available:
- AKCEA-ApoCIII-LRx, an antisense medication; and
- ARO-APOC3, an siRNA to ApoC-III.
Finally, pemafibrate is a fibrate medication that can act as an agonist to PPAR-., reducing triglyceride levels more effectively and with fewer side effects than traditional fibrates. Kowa Pharmaceuticals recently announced they are stopping their study of pemafibrate, as cardiovascular outcomes are unlikely to be met; they may still consider it to be studied for non-alcoholic fatty liver disease/nonalcoholic steatohepatitis (NAFLD/NASH). Clinicians may soon have multiple other options to reduce ASCVD risk in patients with hypertriglyceridemia, but we await the results of trials and updates of guidelines.
11
SUMMARY
For the majority of patients with dyslipidemia, emphasis should first be placed on diet and exercise; causes of secondary hypertriglyceridemia, such as medications or underlying health conditions, should be addressed and augmented as appropriate. Medication therapy may be appropriate for those with triglycerides less than 500 mg/dL if they meet criteria and is generally recommended when values are above 500 mg/dL to prevent pancreatitis. For those who may have resistant hypertriglyceridemia, or suspicion of genetic disorders, referral to a lipid clinic is recommended.
The major challenge, of course, for use of cholesterol-lowering drugs is the problem of long-term non-adherence. Strategies to improve treatment adherence, which honor the patient’s desires and wishes and which are supported by the entire health care team, will give our patients the best chance for more favorable long-term health outcomes.
1
REFERENCES
1. Rosenson RS, Shaik A, and Song W. New therapies for lowering triglyceride-rich lipoproteins: JACC focus seminar 3/4.
J Am Coll Cardiol. 2021;78(18):1817-1830.
2. Takaeko Y, Maruhashi T, Kajikawa M, et al. Lower triglyceride levels are associated with better endothelial function.
J Clin Lipidology. 2021;15:500-511.
3. Aberra T, Peterson ED, Pagidipati NJ, et al. The association between triglycerides and incident cardiovascular disease: what is “optimal”?
J Clin Lipidology. 2020;14:438-447.
4. Miller M, Stone NJ, Ballantyne C, et al. Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association.
Circulation. 2011;123:2292-2333.
5. Virani SS, Morris PB, Agarwala A, et al. 2021 ACC Expert consensus decision pathway on the management of ASCVD risk reduction in patients with persistent hypertriglyceridemia.
J Am Coll Cardiol. 2021;78(9):960-993.
6. Stroes E, Moulin P, Parhoher KG, Rebours V, Lohr JM, Averna M. Diagnostic algorithm for familial chylomicronemia syndrome.
Atherosclerosis Supplements. 2017;23:1-7.
7. Orringer CE, Jacobson TA, and Maki KC. National Lipid Association scientific statement of the use of icosapent ethyl in statin-treated patients with elevated triglycerides and high or very-high ASCVD risk.
J Clin Lipidology. 2019;13(6):860-872.
8. Jacobsen TA, Ito MK, Maki KC, et al. National lipid association recommendations for patient-centered management of dyslipidemia: part 1 — full report.
J Clin Lipidology. 2015;9(2):129-169.
9. Wilson P, Polonsky TS, Miedema MD, Khera A, Kosinski AS, Kuvin JT. Systematic Review for the 2018 AHA/ACC/AACVPR/AAPA/ ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines.
Circulation. 2019;139:e1144-e1161.
10. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines [published correction appears in J Am Coll Cardiol. 2019;73(24):3234-3237].
J Am Coll Cardiol. 2019;73(24):3168-3209.
11. Kowa Research Institute. Kowa to discontinue K-877 (pemafibrate) “prominent” cardiovascular outcomes study. April 8, 2022. Accessed April 28, 2022.
https://www.prnewswire.com/news-releases/kowa-to-discontinue-k-877-pemafibrate-prominent-cardiovascular-outcomes-study-301520956.html