Spring 2026 - Vol. 21, No. 1
SCIENTIFIC REPORT
Case Report of Acute Inflammatory Demyelinating Polyneuritis
Induced by Pembrolizumab
L. Dumasia S. Dumasia Walia
Lena Dumasia, MD
Hematologist/Oncologist, Lancaster Cancer Center
Sonia Dumasia
Student, Manheim Township High School
Anurag Walia, MD, MBBS
Neurohospitalist, Penn State Health
A 61-year-old male presented to his dermatologist in November 2022 with a left shoulder/deltoid lesion that had been present for six months. The left axilla did not reveal any clinically enlarged lymph nodes on exam. He had a biopsy on November 8, 2022, at his dermatologist’s office, and the pathology was consistent with a malignant melanoma with Breslow depth of 4 mm, Clark’s level IV. He had greater than 5 mm2 mitosis.
On December 8, 2022, his plastic surgeon performed a wide local excision of the left shoulder lesion with a sentinel lymph node biopsy. The final pathology revealed a 4.1 mm Breslow depth, and he had 2/2 positive lymph nodes on sentinel lymph node biopsy; thus, he was designated as pT4aN2b, stage IIIC.
Considering his pathology (see Figs. 1 and 2, and Table 1), the patient was referred to the medical oncology service on December 27, 2022, for additional recommendations. His PET scan and brain MRI were negative for metastatic disease.
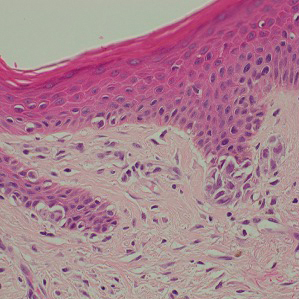

Fig. 1 (left). Hematoxylin-eosin stain of skin depicting malignant melanoma at 40X.
Fig. 2 (right). Hematoxylin-eosin stain of lymph node depicting malignant melanoma at 20X.

Per National Comprehensive Care Network (NCCN) guidelines, the oncology service recommended adjuvant immunotherapy in the form of nivolumab or pembrolizumab for one year due to the high risk of recurrence.
1 Therefore, the patient was started on pembrolizumab on December 30, 2022.
Shortly after initiation, testing revealed that his tumor specimen was positive for the BRAF V600E mutation, suggesting he might benefit from an alternative treatment with BRAF-directed oral therapy in the form of dabrafenib and trametinib. However, because he had already begun treatment with and was having no side effects from the initial pembrolizumab, together the care team and the patient elected to continue the course with pembrolizumab.
At the time of his seventh treatment with pembrolizumab in June 2023, the patient noted he had been experiencing dry mouth over the course of the prior three weeks, rating it a 5/10 in severity. While he did receive pembrolizumab at that time, when he returned three weeks later, he reported the xerostomia had gotten worse, rating it 7/10 in severity at that time.
Hypothesizing that his xerostomia was secondary to autoimmune parotid gland inflammation induced by his immunotherapy, the care team recommended pembrolizumab be held, and a course of corticosteroids was prescribed. He completed a slow taper of prednisone over the course of four weeks and reported a complete resolution of his symptoms at his office visit on July 10, 2023. At that time, he received his eighth dose of pembrolizumab.
Shortly thereafter, he reported a recurrence of his xerostomia, and he began another month of a steroid taper. Pembrolizumab was thus aborted as per protocol regarding immunotherapy-related adverse event recommendations.
2 He had received a total of eight doses.
A few weeks later, on August 8, 2023, he started on standard doses of adjuvant dabrafenib and trametinib, oral BRAF inhibitors. Unfortunately, he experienced side effects from these medications, including fevers and chills, requiring treatment breaks and dose modifications for several months.
In late November 2023, he was diagnosed with uveitis by his ophthalmologist, presumed to be a potential late-occurring side effect of the pembrolizumab, and he was treated with prednisone eye drops.
He did not take his oral BRAF inhibitors during the remainder of November and then restarted dabrafenib and trametinib at the lowest recommended doses in late December 2023. After taking these doses for a few weeks, he developed severe neuropathy of his hands and feet and reported “burning nerve pain” beginning in early January 2024.
On January 8, 2024, he was seen in our office and told to stop taking the oral BRAF inhibitors permanently; the patient was started on gabapentin. However, he continued to have progressive worsening pain in his legs and numbness of the hands and feet.
On January 15, 2024, he presented to an appointment at the oncology office in a wheelchair, his symptoms so severe he was unable to walk. Due to extreme neuropathic pain, we had been titrating his gabapentin and he had begun taking oxycodone, morphine, and lorazepam, yet he was not experiencing sufficient relief at that time.
The patient was directly admitted to Lancaster General Hospital on January 15, 2024, with rapidly progressive ambulatory dysfunction due to weakness and paresthesia. He also had symptoms in the upper extremities. The patient did not have any bulbar dysfunction, dysphagia, speech difficulty, ptosis, or diplopia.
The neurology service was consulted. Their questioning further revealed no history of recent viral infection, recent receipt of vaccines, alcohol use, diabetes, nutritional deficiency, or systemic vasculitis.
On exam, his speech was normal without any aphasia noted. There was no facial weakness, nor ophthalmoplegia. Deep tendon reflexes were absent at the knees, ankles, and triceps but present at both biceps. There was no bulbar dysfunction, dysautonomia, respiratory failure, or speech disturbances.
The motor exam revealed a bilateral foot drop and distal weakness in both lower and upper extremities, with his hand grip judged to be weak bilaterally. His proximal strength across both shoulders and hips was 3+ to 4+/5. On sensory exam, he had impaired fine touch, including sense of vibration, and pain seemed to be manifesting distally in a glove-and-stocking distribution, with the lower extremities more affected than the upper.
The neurology service team suggested that the differential diagnosis included acute inflammatory demyelinating polyneuritis (AIDP), a central lesion such as a brainstem stroke, cervical myelopathy, or thoracic myelopathy.
An MRI of the brain and spine did not reveal overt pathology, and there were no findings from imaging studies to explain his neurologic and muscular concerns. He also had a bone scan that did not reveal any evidence of new or recurrent cancer.
The patient underwent a lumbar puncture on January 16, 2024 (see Table 2).

The neurology service concluded that his diagnosis was most likely AIDP, also known as Guillain-Barré syndrome (GBS).
Due to his rapidly progressive course, and with a working diagnosis of GBS, the neurology service team recommended treatment with intravenous immunoglobulin (IVIG), and the patient accepted the risks and agreed to proceed with the treatment. He received IVIG 400 mg/kg per day for five days, as well as physical therapy, occupational therapy, and speech therapy.
He was discharged from Lancaster General Hospital on January 23, 2024, and sent to an outpatient rehabilitation facility. Unfortunately, he required routine doses of morphine, gabapentin, duloxetine, trazodone, and oxycodone as needed after discharge from rehab to his home to help control his pain. He required a wheelchair and then a walker for many months afterward.
Finally, after several months of therapy, his weakness had nearly resolved, and he was back to his favorite hobby of biking in October 2024. He continues, however, to require prescription pain medications, from which he is still trying to wean. His latest scans reveal that his cancer remains in remission.
DISCUSSION
Pembrolizumab is a humanized monoclonal immunoglobulin antibody that binds to the program cell death 1 (PD-1) receptor and prevents interaction with the program death-ligand 1 (PD-L1) and program death-ligand 2 (PD-L2).
3 Somatic cells produce these ligand proteins that attach to the PD-1 receptor on T cells and induce inhibitory signals, causing T-cell deactivation.
2,4 This process negatively regulates the immune system to prevent autoimmunity; the PD-1 receptor may thus be considered an immune checkpoint.
3
Tumor cells often upregulate PD-L1 and PD-L2 ligands on their cell surfaces to evade T-cell immunity. By inhibiting this interaction, pembrolizumab preserves our T cell-mediated immune response, which can result in attack and destruction of tumor cells.
4,5
PD-L1 and PD-L2 expressions have been found in many tumors, including melanoma, ovarian, lung, and renal cell carcinomas.
3
Clinicians must be aware of all potential immune-related adverse events that can happen to an individual under the immuno-oncology approach.
3 AIDP, as an adverse result of treatment with checkpoint inhibitors, has not been frequently described in the medical literature.
6-12 It is critical to immediately recognize this complication, as checkpoint therapy must be permanently discontinued and treatment must be promptly delivered.
3
This case highlights the rare but serious risk of AIDP associated with immune checkpoint inhibitor therapy, such as pembrolizumab, in patients with high-risk melanoma. Early recognition and prompt intervention with immunomodulatory therapies, including IVIG and/or steroids, are critical for optimizing patient outcomes.
13,14
Any new or worsening neurological symptom in a patient receiving an immunotherapy agent — regardless of the diagnosis — should prompt an immediate evaluation by the treatment team and consideration of consultation with the neurology service.
3 Clinicians must be keen to recognize when AIDP is a result of checkpoint therapy, as a misdiagnosis or delay in treatment could be fatal.
3
Early intervention can potentially prevent complications such as respiratory failure and death.
2 More research is needed to understand the development of these rare iatrogenic outcomes, including the risk factors that may predispose cancer patients, so that efforts might focus on prevention.
3
ACKNOWLEDGEMENT
Special thanks to Grace W. Weyant, MD, from LGHP Pathology, who helped prepare the photos in Figs. 1 and 2.
REFERENCES
1. NCCN Guidelines for Patients: Melanoma. National Comprehensive Cancer Network; 2024.
2. Keytruda.
® n.d. Accessed January 12, 2026.
https://www.keytruda.com/hcp/
3. Manam R, Martin JL, Gross JA, et al. Case reports of pembrolizumab-induced acute inflammatory demyelinating polyneuropathy.
Cureus. 2018;10(9):e3371.
4. Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity.
Immunol Rev. 2010;236:219-242.
5. Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy.
Nat Rev Cancer. 2012;12(4):252-264.
6. Naidoo J, Page DB, Li BT, et al. Toxicities of the anti-PD-1 and anti-PD-L1 immune checkpoint antibodies.
Ann Oncol. 2015;26(12):2375-2391.
7. Day D, Hansen AR. Immune-related adverse events associated with immune checkpoint inhibitors.
BioDrugs. 2016;30(6):571-584.
8. Zimmer L, Goldinger SM, Hofmann L, et al. Neurological, respiratory, musculoskeletal, cardiac and ocular side-effects of anti-PD-1 therapy.
Eur J Cancer. 2016;60:210-225.
9. Kao JC, Liao B, Markovic SN, et al. Neurological complications associated with anti-programmed death 1 (PD-1) antibodies.
JAMA Neurol. 2017;74(10):1216-1222.
10. Liao B, Shroff S, Kamiya-Matsuoka C, Tummala S. Atypical neurological complications of ipilimumab therapy in patients with metastatic melanoma.
Neuro Oncol. 2014;16(4):589-593.
11. Sepúlveda M, Martinez-Hernandez E, Gaba L, et al. Motor polyradiculopathy during pembrolizumab treatment of metastatic melanoma.
Muscle Nerve. 2017;56(6):162-167.
12. Astaras C, de Micheli R, Moura B, Hundsberger T, Hottinger A. Neurological adverse events associated with immune checkpoint inhibitors: diagnosis and management.
Curr Neurol Neurosci Rep. 2018;18(1):3.
13. Loo J, Yip S, Tan S. Pembrolizumab-induced Guillain-Barré syndrome in stage III melanoma: a case report and review of the literature.
World J Clin Oncol. 2025;16(2):978-987.
14. Piccione EC, Rytlewski JA. Neurologic complications of immune checkpoint inhibitors: Guillain-Barré syndrome.
Front Immunol. 2020;11:1132.