
Fall 2019 - Vol. 14, No. 3
Reducing Opioid Overdose Deaths
in Lancaster County:
The Rise of an Epidemic, and LGH's Response
Tara M. Tawil, M.D.
Lead Physician, Penn Medicine Lancaster General Health
Drug and Alcohol Task Force
Physician Advisor for Case Management at LGH
BACKGROUND
There were 168 drug-related overdose deaths in Lancaster County in 2017. This startling statistic is just the local manifestation of a national epidemic that has made opioids the leading cause of death for Americans under the age of 50. Since 1999, opioids have claimed the lives of more than 700,000 Americans; currently this amounts to an average of 130 every day. Between 2016 and 2017, Pennsylvania experienced a 43% increase in opioid overdose deaths, the highest statewide increase in the country.1,2
HOW DID THE OPIOID EPIDEMIC DEVELOP?
The Centers for Disease Control (CDC) identifies three separate waves of this epidemic (Fig. 1):

Fig. 1. Three waves of the rise in opioid overdose deaths. Source: National Vital Statistics System mortality file
The first wave began with an increase in the prescription of opioids in the 1990s, which reflected changes in the promotional practices of drug manufacturing and retailing companies,* and in the prescribing routines of physicians. This wave provoked statewide drug monitoring programs, and CMS programs that tracked the prescribing records of physicians. Though these actions caused the number of prescriptions to decrease, a second wave developed even as opioid prescriptions began to plateau and decline.
The second wave began in 2010, with a rapid increase in deaths related to heroin. It has been speculated that when providers were challenged on their prescribing practices, some patients were not tapered off opioid therapy appropriately, and/or did not have their underlying addiction addressed. In my personal experience as a hospitalist, I began to see firsthand an increase in complications from the use of illicit drugs, including IV drugs. Some of my patients described attempting to buy prescribed opioids off the street, and — finding them too expensive — turned to heroin.
The third wave started in 2013 when there was an increase in the manufacture of synthetic opioids, particularly illicit fentanyl. In this wave not only did the potency of readily available opioids increase, but new combinations of drugs became available, such as cocaine and marijuana laced with illicitly manufactured fentanyl. When people who had not acquired a tolerance for high potency opioids were exposed to these more potent drugs, as well as to combinations that hid a potentially lethal component, there was a rapid rise in fatal accidental overdoses.
OPIOID ADDICTION AND ITS MANAGEMENT IN THE 18th AND 19TH CENTURIES
Attempts to understand illicit drug and alcohol abuse go back centuries, and the situation at the time of the American Revolution was documented by colonial civic leader and Philadelphia physician Dr. Benjamin Rush. His 1784 pamphlet “An Inquiry into the Effects of Ardent Spirits on the Human Mind and Body” focused on the conditions that result from excessive alcohol use, and discussed which patients might be more susceptible to them. He described alcoholism as a progressive disease, and his views on abstinence shaped some of the views on alcohol use disorder that persist to this day. Interestingly, he mentioned opium as a potential substitute for alcohol, while reflecting on its misuse as well.3,4
During the Civil War the Union Army alone issued nearly 10 million opium pills and 2.8 million ounces of opium powder and tinctures. The hypodermic syringe introduced in 1856 led to use of injectable opioids. By the end of the 19th and beginning of the 20th century, morphine and opium powders were widely used by the general population. In 1888, opiates made up 15% of all prescriptions dispensed in Boston.3 The overuse of opiates peaked in one of the first opiate epidemics in 1895 (Fig. 2).

Fig. 2. Opioid addiction was a common problem at the end of the 19th century.
Medical journals through the 1880s and 1890s started to publish more information on the risk of opioid addiction, and medical schools became more aggressive in educating about the risk of opioids. Laws regulating opioids were passed between 1895 and 1915, followed by a distinct reduction in their prescription and use.
THE PATHOPHYSIOLOGY OF ADDICTION
Addiction was initially a contested term, and it was first used as a medical term in the early 20th century. At that time, it was often viewed as a moral failure resulting from a voluntary decision. Only recently has our understanding of the neurobiology of motivation, and the lasting impact of addictive drugs on the brain, led to the mainstream view that addiction is a chronic relapsing disorder characterized by a compulsive drive to take drugs despite adverse consequences.
First, it is important to differentiate between physical dependence and addiction. Physical dependence results from drugs that cause short-term withdrawal symptoms if they are removed. Addiction refers to the structural and functional disruption of the regions in the brain that control motivation, emotional regulation, inhibitory control, and awareness.

Fig. 3. The three stages of addiction cycle and the brain
regions associated with them. Image from Chapter 2 of
Facing Addiction in America: The Surgeon General's
Report on Alcohol, Drugs, & Health (Internet),
November 2016.6
The three stages of addiction areas are frequently described as a feedback loop (Fig. 3): 5
1) The “binge-intoxication phase” is the initial incentive that has its main impact in the basal ganglia. Essentially it is the “high” from taking a drug, and can be directly tied to the environment or situation.
2) The next stage is the “withdrawal–negative effect” stage which occurs in the extended amygdala and habenula. This is where symptoms of stress and reward deficit exist, and this stage may be accompanied by dysphoria and irritability.
3) The last stage is known as the “preoccupation-anticipation” stage. It is centered in the prefrontal cortex which houses our impulses and our ability to maintain control. Damage and change in the metabolism of crucial portions of the brain, such as the prefrontal cortex, can result in disruption of inhibitory control and a return to the binge-intoxication stage despite adverse consequences. This stage directly affects the executive functioning of the brain. This feedback loop can result in abnormal behaviors that persist after the cessation of drug use, which is why addiction is often described as a chronic disease.5
It is crucial to note that the frontal lobes and their connections are not fully formed until the age of 25. Abundant information in the literature suggests that early drug use before these connections are complete is associated with greater vulnerability to addiction, and addiction that is more difficult to treat.7 Since the first exposure to drugs often occurs in adolescence, it is vital to focus on prevention and support for teens.
PROGRESS IN RECOGNITION AND TREATMENT OF OPIATE ADDICTION
Initial programs for treatment were broken down into three approaches: a) rapid and complete withdrawal; b) gradual withdrawal using a stepdown of drug dosage over time; and c) prolonged withdrawal over an extended period of time with a wide range of treatments. In the late 19th and early 20th century withdrawal protocols were varied and included the Lambert-Towns treatment (a combination of belladonna, xanthoxylun, digitalis, hyoscyamus, and strychnine); the Sceleth method (scopolamine, pilocarpine, ethyl-morphine, and strychnine); and Narcsan treatment (lipoids, proteins and vitamins). During the early 20th century, the use of medicinal substances to treat withdrawal symptoms and addiction increased, and included use of opium, cocaine, morphine, and codeine. Morphine maintenance clinics were also seen from 1919-1925 but were threatened with federal indictment.4
The next large breakthrough was methadone (dolophine), a long acting synthetic narcotic initially developed in Germany in 1943, and used for maintenance in the United States beginning in the 1960s. Drs. Senay and Renault in 1971 showed significant improvement with methadone, including reductions in drug use, criminal activity, and health-related problems, as well as increased rates of employment, and improvement in social and psychological health.8 Use of methadone increased particularly in the 1970s during the post-Vietnam War era.
The first narcotic antagonists were introduced in 1941, including nalorphine (Nalline), followed by naloxone and naltrexone, and were first used clinically in the 1960s. When studied by Senay and Renault in 1971, their usage was limited by side effects and the need for patient motivation. In the 1980s, narcotic antagonists began to gain favor in specific populations such as opiate-addicted physicians.
Clonidine was identified in 1980 for treatment of a wide range of opiate withdrawal symptoms. Though it did not cause sedation or intoxication, doses were limited by the side effects of bradycardia and hypotension; these made clonidine useful for short-term management of withdrawal, but not practical for titrating doses to manage further craving.
Buprenorphine is a partial agonist with antagonistic properties; it was approved by the FDA in 2002, resulting in a new era of modern treatment which is currently being used in our clinics.9 It has a higher affinity for the mu opioid receptor than other opioids, but a plateau effect makes it easier to titrate doses and patients can administer it at home. This property has ushered in an era of treatment for addiction that can take place in a primary care office.
IDENTIFICATION/DIAGNOSIS
The most commonly used practice pathway for identification of addiction is known as “Screening, Brief Intervention, and Referral to Treatment” or SBIRT. This is an integrated public health approach to early intervention for people with substance use disorders that uses standardized screening tools to detect risky behaviors that lead to substance use.10 The standardized screening tool can be simple (e.g. a single screening question), or more wide-ranging, such as NIDAMED, a comprehensive tool developed by the National Institute on Drug Abuse (NIDA) in 2009. It includes screening for tobacco, alcohol, illicit drug, and nonmedical prescription drug use.
NIDA-Modified ASSIST is an abbreviated tool to lead into the ASSIST** questionnaire which reviews substance use across a lifetime and can assess the risk of further use. DAST-10 (Drug Abuse Screening Tool 10) is a 10 item yes/no self-report instrument that was condensed from the 28 item DAST. It should take less than eight minutes to complete, and was designed to provide a brief instrument for clinical screening.11
Some screening tools have focused on special populations.
1) CRAFFT, which is a mnemonic acronym of the first letters of key words in the form’s six screening questions, has been clinically validated for use in the adolescent population from ages 12-18.
2) The 5Ps screen for prenatal drug and alcohol use is an effective tool for engagement with pregnant women.12
3) AUDIT PC and single substance use screening questions for 12 months can serve as an abbreviated way of assessing drug and alcohol use in the hospital setting.13
The role of any screen is to identify not only patients who have a substance use disorder but those who may be at risk for developing one. Screens offer a unique opportunity for a physician or advanced practice provider to have a discussion regarding the medical consequences of use, as well as providing a chance to reduce potential harm. For example, for those abusing opioids the discussion is a crucial opportunity to reduce the risk of a fatal overdose by giving a naloxone prescription, and identifying the reasons for opioid misuse that may be treatable, such as pain.
TREATMENT MODELS AND THE CONTINUUM OF CARE
It is now recognized that many patients with substance use disorders have concurrent chronic health conditions. Also, if patients are treated “successfully” and return to their families and jobs, they may face the same environment that originally resulted in drug use, and it can continue to be a trigger for relapse and reuse. Therefore, the focus of care for substance use disorders has shifted away from treatment isolated to specific programs, and toward a wider spectrum of more comprehensive treatment options.

Fig. 4. The Continuum of Care Model developed by the Substance Abuse
and Mental Health Services Administration (SAMHSA).
The Continuum of Care model (Fig. 4) was developed as a comprehensive approach by the Substance Abuse and Mental Health Services Administration (SAMHSA) to integrate promotion of general health, prevention and intervention for drug abuse, as well as support for sustained recovery. SAMHSA currently advocates for integrated care, which leads to better health outcomes when structured care addresses physical and behavioral issues in the same place. This approach also highlights the importance of approaching addiction as a disorder that requires not only identification and treatment, but also reduction of risk by behavior modification.
The first step in addiction management is to provide an initial level of care assessment by a drug and alcohol certified counselor or medical provider.14

Fig. 5. Five levels of assessment help determine the level of care based on patient needs.
Source: American Society of Addiction Medicine 2013
The five levels of assessment (Fig. 5) include:
a) level 0.5: early intervention and education;
b) level 1: outpatient services;
c) level 2: intensive outpatient and partial hospitalization services;
d) level 3: residential/inpatient services;
e) level 4: medically managed intensive inpatient services.
Within these five levels of care, there should be a continuum for patients based on need.
Treatment of substance abuse disorders now commonly involves Medication-Assisted Treatment (MAT) using one of three FDA-approved medications in combination with counseling and behavioral therapies. Methadone, naltrexone, and buprenorphine have all yielded improved continuity of care, reduced relapse rates, and fewer deaths from opioid overdoses compared with no medication.15,16
Current treatment models also incorporate partnerships with primary care and behavioral health. These models have helped reduce the isolation of patients receiving addiction care, since they can now receive it in the same place where they receive general medical care. This model of integrated care is the framework for the way Penn Medicine LGH approaches opioid use disorder (see below).
Prior to 2017, Penn Medicine Lancaster General Health had limited ability to provide the first level of care through outpatient services because there weren’t enough physicians to do so, and care did not include integrated counseling support. In 2017, Penn Medicine Lancaster General Health was awarded a Centers of Excellence grant to implement integrated counseling into primary care practices, and to partner with community providers for specialty mental health services and treatment of substance use. This program allowed primary care providers to participate in MAT with buprenorphine. Prior to 2017 only four providers were able to provide MAT with buprenorphine in one clinic through Lancaster General Health,9 whereas there are now more than 30 providers with the training and ability to provide it in 13 LGHP family medicine practices across the county.
In November of 2018, we also attested to a hospital quality improvement program released by the Department of Health and Hospital Association of Pennsylvania for follow up after treatment in the emergency department. With the addition of 24-hour coverage in the emergency department of dedicated case management for substance and alcohol-related issues, we were now able to offer a higher level of care assessment, and to better meet patients’ needs. This expansion of coverage provided access to a whole new group of patients (Fig. 6), since the ED is where one-third of patients with substance use disorders have their first contact with the medical community. It also allowed us to offer additional outpatient services to patients where they receive their primary care.

Fig. 6. A Centers of Excellence grant in 2017 enabled Penn Medicine Lancaster General Health to offer services to an increasting number of patients.
EDUCATION AND PREVENTION IN LANCASTER COUNTY
In 2017, Lancaster County Joining Forces was launched as a community collaboration with Penn Medicine Lancaster General Health to coordinate countywide efforts to reduce opioid-related deaths through education. This initiative focused on saturating the community with information, increasing access to treatment and recovery resources, and spreading the use of the drug naloxone to reduce the incidence of opioid overdoses. The model focuses on promotion of well-being and the ability of individuals to withstand challenges.
The program’s website (www.lancasterjoiningforces.org) advocates universal prevention, and provides educational tools such as materials, toolkits, and brochures. School-based prevention curricula allow health care professionals to provide information to children and teens. Lancaster Joining Forces also united with the Lancaster County Anti-Heroin Task Force, which was created by local mayors to provide county forums as an interface between local government, law enforcement, and Lancaster County residents.
Penn Medicine Lancaster General also plays a vital role in the South Central Opioid Awareness Coalition, which brings together health systems, hospitals, health care providers, and pharmacists to address opioid addiction through awareness and education. The coalition provides standardized messaging to be used by health systems, pharmacists, and community-based organizations. It also assembles evidence-based resources from multiple health system partners and supporters, including Wellspan, Geisinger, Penn State, UPMC Pinnacle, and Summit Health.
THE LOCAL EXPERIENCE
In Pennsylvania, the increase in substance abuse was reflected in a rise in the incidence of fentanyl seizures from fewer than 1,000 in 2014 to more than 30,000 in 2016.2 In Lancaster County, the number of drug-related overdose deaths rose 180% between 2014 and 2017.17
As a result of the advances in 2017, with formation of Lancaster Joining Forces and receipt of the Centers of Excellence grant, use of Medication-Assisted Treatment increased substantially. Between 2017 and 2018, opioid deaths in Lancaster County fell from 168 to 108 annually (Fig. 7). 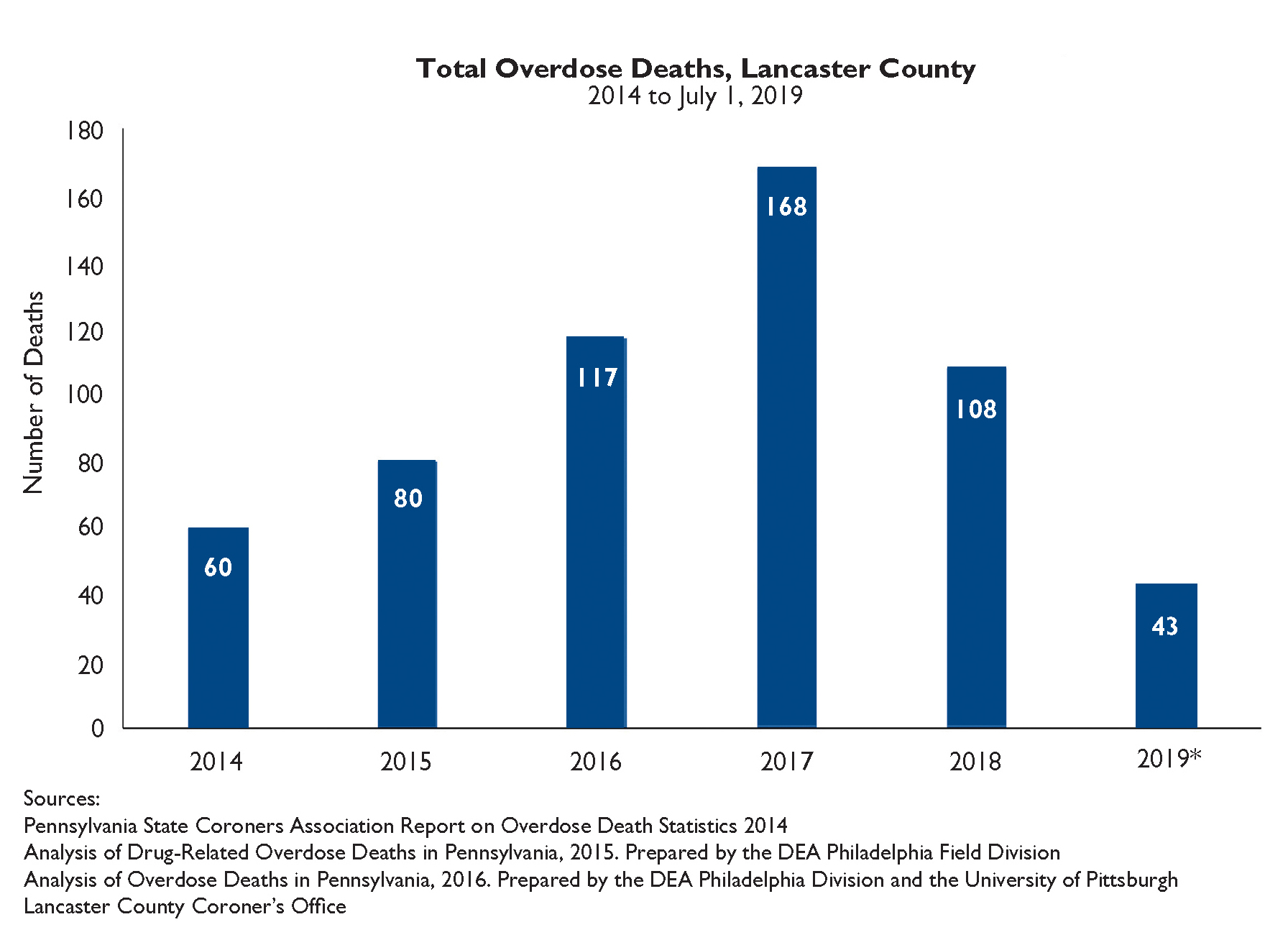
Fig. 7. Total overdose deaths in Lancaster County dropped from 168 in 2017, to 108 in 2018.
* Deaths as of July 1, 2019
Though this was still twice as high as it had been in 2014, Lancaster County was one of only three counties in the state of Pennsylvania where overdose deaths declined in 2018. This trend can continue, but only if physicians and the entire community have a clear understanding of the prevention, diagnosis, and treatment of addiction.
CONCLUSION
In the last two years, Lancaster County has been one of only three counties in Pennsylvania where the number of opioid-related overdose deaths has fallen. By integrating care for substance abuse into primary care, as well as shifting the focus of management from episodic interventions to a continuum of care that extends from prevention to recovery and may include medication management, we may be on the threshold of a seismic change.
If we consider addiction a chronic disease, and we break down the barriers to accessing treatment, we can hopefully anticipate a future of successful long-term management of Opioid Use Disorder.
* In July 2019, thousands of confidential documents from corporations in the pharmaceutical and retail industries were unveiled in federal court filings in Cleveland, OH. Lawyers for cities, towns, and counties are seeking billions of dollars in compensation from corporations implicated in the opioid epidemic. According to the filings, those responsible for feeding the epidemic were not just familiar players like Purdue Pharma, but also generic manufacturers, superstores like Walmart, and chain retailers like Rite Aid, who flooded the country with billions of pills.
** ASSIST: Alcohol, Smoking, and Substance Involvement Screening Test (originally developed by the World Health Organization).
REFERENCES
1. CDC National Opioid data. www.cdc.gov/opioids/strategy
2. Opioid Threat in PA. Joint Intelligence Report DEA-PHL-DIR September 2018
3. Trickey E. Inside the story of America’s 19th cetury opioid addiction. Smithsonian.com; January 4, 2018. https://www.smithsonianmag.com/history/inside-story-americas-19th-century-opiate-addiction-180967673/
4. White WL and Webber R. History of pharmacologic treatment for substance use disorders. La Historia de la Psicofarmacologia. 2006. Madrid: Editorial Médica Panamericana, pp. 887-898.
5. Koob G and Volkow N. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psych. 2016; 3(8): 760-773.
6. https://www.ncbi.nlm.nih.gov/books/NBK424849/figure/ch2.f3
7. King KM and Chassin L. A prospective study of the effects of age of initiation of alcohol and drug use on young adult substance dependence. J Studies Alc Drugs. 2007; 68(2): 256-265.
8. Senay EC, Renault PF. Treatment methods for heroin addicts: A review. J Psychedelic Drugs. 1971; 3(2): 47-54. http://dx.doi.org/10.1080/02791072.1971.10471376
9. Paist SS. Use of buprenorphine in the management of opiate addiction. J Lanc Gen Hosp. 2012; 7(4):114-116
10. Babor RF, Del Boca F, Bray JW. Screening, brief intervention and referral to treatment: implications of SAMHSA's SBIRT initiative for substance abuse policy and practice. Addiction. 2017; Feb: 112 (Suppl 2): 110-117
11. Bohn MJ, Babor TF, Kranzler HR. Validity of the drug abuse screening test (DAST-10) in inpatient substance abusers. Problems of Drug Dependence 1991. Proceeding of the 53rd Annual Scientific Meeting of the Committee on Problems of Drug Dependence, Inc. https://archives.drugabuse.gov/sites/default/files/monograph119.pdf#page=263
12. Chasnoff IJ, Well AM, McGourty RF, et al. Validation of the 4P's Plus screen for substance use in pregnancy. J Perinatol. 2007; 27 (12): 744-48.
13. Pecoraro A, et al. Using the AUDIT-PC to predict alcohol withdrawal in hospitalized patients. J Gen Int Med. 2014; 29 (1): 34-40.
14.; The ASAM Criteria: Treatment criteria for addictive, substance, related and co-occurring conditions. 2013. Published by the American Society for Addiction Medicine. https://www.asam.org/resources/the-asam-criteria
15. Kakko J, Svanborg KD, Kreek MJ, et al. 1-year retention and social function after buprenorphine-assisted relapse prevention treatment for heroin dependence in Sweden: a randomized, placebo-controlled trial. Lancet. 2003; 361:662-668.
16. Larochelle MR, Bernson D, Land T, et al. Medication for opioid use disorder after nonfatal opioid overdose and association with mortality: a cohort study. Ann Int Med 2018; 169(3):137-145. doi: 10.7326/M17-3107.
17. Lancaster County Coroner’s Office Data. March 2019.